|
8/10/2023 0 Comments Cu electron configuration 
In nature it has two isotopes, 63 (69.09%), which has 29 electrons and protons and 34 neutrons, and 65 (30.91%), which has 29 electrons and protons and 36 neutrons. Copper was one of the earliest known metals, having reportedly been mined for over 5000 years. Copper (Cu) has two valences Cu I (cuprous) has one valence electron and Cu II (cupric) has two valence electrons. Copper typically forms a bluish green solution. 
Like many of the transition elements, copper has a colored ion. In addition, it has more than one oxidation state. It is one of the transition elements that actually uses electrons from one of the inner orbitals in chemical reactions. Additional Notes: Copper is a very interesting element.The skin of the Statue of Liberty is made of copper. Its alloys are used in jewelry, bronze sculptures and for coins. Uses of Copper: Most often used as an electrical conductor.Primary mining areas are in USA, Zaire, Zambia, Canada, Chile, Cyprus, Russia and Australia. Annual world production is around 6,540,000 tons. Copper is obtained by smelting, leaching and by electrolysis. 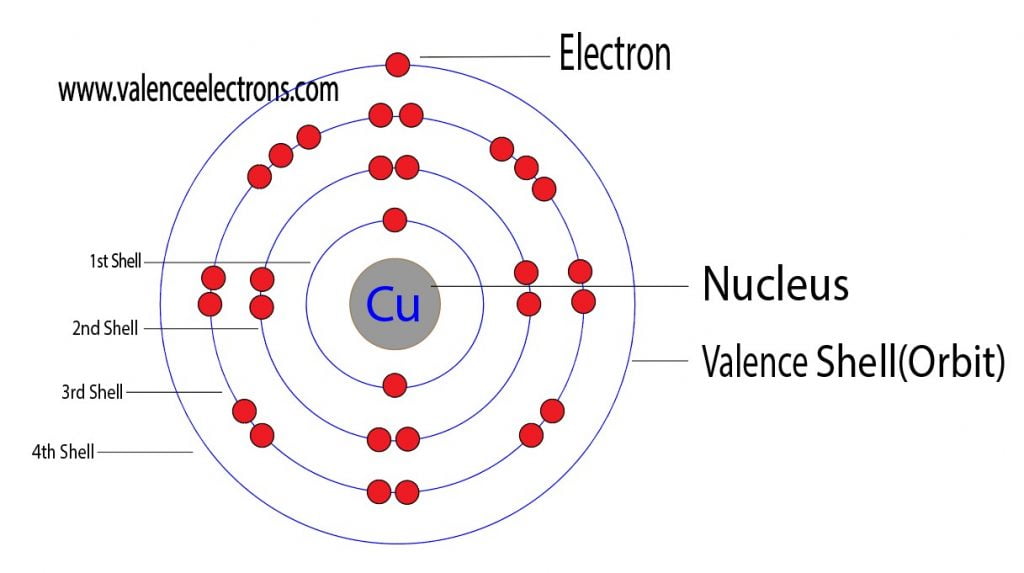
Usually copper found in such minerals as azurite, malachite and bornite and in sulfides as in chalcopyrite (CuFeS 2), coveline (CuS), chalcosine (Cu 2S) or oxides like cuprite (Cu 2O). Sources of Copper: Pure copper occurs rarely in nature.Name Origin: Latin: cyprium (island of Cyprus famed for its copper mines).Discoverer: Known to ancient civilization.70kg human: 72 mg Who / Where / When / How Note: this data represents naturally occuring levels of elements in the typical human, it DOES NOT represent recommended daily allowances. Target Organs: Eyes, skin, respiratory system, liver, kidneys (increase(d) risk with Wilson's disease). 
Routes of Exposure: Inhalation Ingestion Skin and/or eye contact.Vapor Pressure = C Regulatory / Health.Flammablity Class: Non-combustible solid (except as dust).Enthalpy of Vaporization: 304.6 kJ/mole.Enthalpy of Atomization: 338.9 kJ/mole 25☌.Description: Reddish orange transition metal.Conductivity Electrical: 0.596 10 6/cm Ω.Coefficient of lineal thermal expansion/K -1: 16.5E -6.Valence Electron Potential (-eV): 34 Physical Properties of Copper.Incompatibilities: Oxidizers, alkalis, sodium azide, acetylene.Electronegativity: 1.9 (Pauling) 1.75 (Allrod Rochow).Electrochemical Equivalent: 1.1855g/amp-hr.Valence Electrons: 3d 10 4s 1 Chemical Properties of Copper.Number of Neutrons (most common/stable nuclide): 35.Number of Electrons (with no charge): 29.Electrons per Energy Level: 2,8,18,1 Shell Model.Electron Configuration: 1s 2 2s 2p 6 3s 2p 6d 10 4s 1.Crystal Structure: Cubic face centered.Cross Section (Thermal Neutron Capture)σ a/ barns : 3.78.Swedish: Koppar Atomic Structure of Copper.Series: Transition Metals Copper's Name in Other Languages.Common Chemical Compounds of Copper Overview of Copper.In addition technical terms are linked to their definitions and the menu contains links to related articles that are a great aid in one's studies. Common chemical compounds are also provided for many elements. Skip to site menu on this page Periodic Table of Elements Element Copper - CuĬomprehensive data on the chemical element Copper is provided on this page including scores of properties, element names in many languages, most known nuclides of Copper. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
